Nitrous Oxide 101: When It Comes to Climate Change It’s No Laughing Matter

You’ve probably heard of nitrous oxide when you’ve visited the dentist for a procedure and been given laughing gas as a mild anesthetic. What you may not know is that nitrous oxide (N₂O) is one of the most potent greenhouse gases (GHGs). The potency of greenhouse gases is measured by their global warming potential (GWP) and compared relative to carbon dioxide (CO₂). GWP measures the ability of an atmospheric gas to absorb energy from the environment, thereby increasing the temperature in the atmosphere. In agriculture, we are most concerned with mitigating the impacts of three GHGs: CO₂, N₂O, and methane (CH₄). The GWP over a 100 year time horizon for each of these are listed below
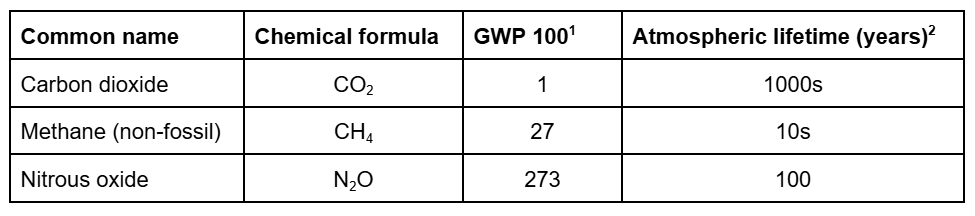
From the chart above we can see that N₂O is over 250 times more potent as a greenhouse gas than CO₂. You may be asking yourself if this is the case, why do we only hear about CO₂ in the context of global warming and climate change? The answer lies in the atmospheric lifetime of each particular gas, i.e. how long does it stay in the atmosphere. Carbon dioxide remains in the atmosphere for thousands of years, with about 25% essentially remaining forever. Therefore, it is imperative to minimize current CO₂ emissions while removing as much previously released CO₂ as possible.
Nitrous oxide has the highest warming potential of the three and persists for a century. This unique combination presents a significant opportunity to slow warming in a relatively short time. The more N₂O emissions we can eliminate now, the more rapidly we can slow warming in the immediate future. The same is also true for CH₄, but that’s a subject for another post. Make no mistake, this is not an either/or proposition. We must reduce the emissions for all three as rapidly as possible.
The Nitrogen Cycle in Agriculture
Nitrogen (N₂) is the most abundant gas in our atmosphere, comprising about 78% of the air we breathe. It is critical for plant growth and development, however the relative chemical stability of N₂ makes it directly unavailable for plant metabolism. This problem was solved in the early 20th century by chemist Fritz Haber who developed the process of converting N₂ into ammonia by reacting it with hydrogen gas and an iron catalyst at high temperature. Ammonia is more chemically reactive and thus available for the biochemical reactions that are essential for plant growth. This process for which Haber was awarded the Nobel Prize in Chemistry in 1918 allowed for the large-scale production of chemical fertilizers. It cannot be understated that this invention played a critical role in helping feed billions of people and sustain communities around the world from the 20th century to today.
However, the increased availability of nitrogen from ammonia that makes it ideal for producing more plant biomass, and thus more food, also creates the undesirable side effect of allowing for its release back into the atmosphere in the form of the greenhouse gas N₂O. Fertilizers on the surface of a field are subject to breakdown by various physical and chemical processes, i.e. air oxidation. Fertilizers contained in the soil are also subject to breakdown through chemical processes as well as the action of soil bacteria. These forms of soil nitrogen are then available to be released into the atmosphere or to leach into groundwater through the soil where they contaminate water supplies or are released back into the atmosphere. In the US, 75% of all N₂O emissions originate from agricultural activities³. This demonstrates the enormous potential for the agriculture sector to positively impact global warming through the reduction of nitrous oxide emissions.
Reducing N₂O Emissions in Agriculture
When it comes to food production, fertilizer application has long been seen as a relatively affordable mechanism to protect crop yields. Given the often thin margins in farming, it is understandable that fertilizer application could be seen as an insurance policy to help maintain farm profitability. This approach can lead to over application of fertilizer, opening the door for release of N₂O into the atmosphere. Fortunately, we have a few simple tools along with some innovations that can help us to reduce the GHG and environmental impacts from the use of fertilizers:
4R Method
One of the most widely adopted approaches to improving fertilizer efficiency and reducing environmental impacts is the 4R nutrient management framework.
- Right source: Match the type of fertilizer to the specific crop so plants can use nitrogen as efficiently as possible.
- Right time: Apply fertilizer as close as possible to when crops need it most to improve uptake and reduce environmental losses.
- Right rate: Apply neither too little nor too much fertilizer—balancing crop needs with yield goals, which is often the most challenging but most impactful step.
- Right place: Apply fertilizer as close as possible to where plants can absorb it, minimizing nitrogen loss to air and water.
By adopting management techniques such as 4R, Project Drawdown estimates that we could realize up to a 76% reduction in N₂O emissions from fertilizer usage.
Innovations in Fertilizers and Alternatives
In addition to the agronomic approach of 4Rs, reducing nitrous oxide (N₂O) emissions in agriculture requires both traditional agronomic strategies and emerging technological innovations.
Enhanced Efficiency Fertilizers (EEFs) are specialized formulations that include inhibitors to slow fertilizer breakdown by environmental reactions or soil microorganisms. Slower breakdown of fertilizers increases nutrient availability to plants while reducing N₂O and other emissions.
Another exciting tool to potentially reduce the impact of fertilizers on N₂O emissions is the adoption of biological products such as Plant Growth-Promoting Rhizobacteria (PGPR) and biostimulants. The application of microbial formulations directly into the field are believed to aid growing plants in the enhanced uptake of nutrients from the soil. Some formulations containing nitrogen-producing microbes may also directly provide nitrogen to the plants. This is still a relatively new field of research in plant nutrient management and like any new innovation, the jury is still out on whether the technology will be widely applicable across many crops and many geographies. Nevertheless, these are the types of innovations that could also play a role in the reduction of N₂O emissions.
HabiTerre’s Approach to Modeling N₂O Mitigation
HabiTerre’s SYMFONI™ platform is powered by ecosys, a mechanistic, process-based model that simulates how nitrogen actually moves through agricultural systems. When grounded in real-world data, process-based models can help companies design effective N₂O reduction strategies and clearly measure the climate benefits of better nitrogen management.
What makes ecosys different from other models is its ability to represent the underlying drivers of nitrogen emissions—such as soil moisture, oxygen availability, and microbial activity—rather than relying on simplified formulas. These factors are essential for accurately estimating N₂O emissions and understanding how management changes reduce them.
This science-first foundation makes SYMFONI the ideal platform to evaluate the climate impact of different fertilizer strategies, including 4R nutrient management, enhanced efficiency fertilizers, and biological products.
Going Forward
Our ability to reduce N₂O emissions in agriculture can have a significant impact on slowing global warming over a relatively short period of time. These emission reductions come not only from losses on the field, but also from eliminating GHG emissions from the fertilizer production itself. The tools we have to do this include agronomic approaches and new innovations. Better nutrient management not only has the potential to reduce N₂O emissions, but these approaches also reduce other environmental harms from nitrogen runoff such as watershed contamination and algal blooms in our lakes.
HabiTerre’s SYMFONI platform is uniquely positioned to assist our partners in accurately estimating the N₂O emissions reductions from their regenerative agriculture programs. Our ecosys model performs at the top of the class for N₂O mitigation estimates as demonstrated by its validation by the Climate Action Reserve. Additionally, our team is actively exploring the integration of the latest innovations in nutrient management into our platforms. Look for those announcements in the coming weeks and please reach out to the team if you have interest in exploring innovative solutions to N₂O mitigation.
¹GWP 100 is taken from the IPCC 6th Assessment Report 2021 (AR6). https://www.ipcc.ch/assessment-report/ar6/
³US Environmental Protection Agency. https://www.epa.gov/ghgemissions/nitrous-oxide-emissions



